Authors:
Altamash Ashfaque1, Caryl Richards2, Zaid Khan1
Affiliation:
1. University Hospitals of Leicester, Leicester, UK.
2. Department of Cardiovascular Sciences, University of Leicester, Leicester, UK.
Case Summary
A 51-year-old man, undergoing routine surveillance following treatment for colorectal malignancy, attended for follow-up imaging. His past medical history included an ischemic stroke, with a prior echocardiogram raising the possibility of apical hypertrophic cardiomyopathy.
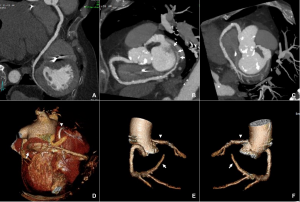
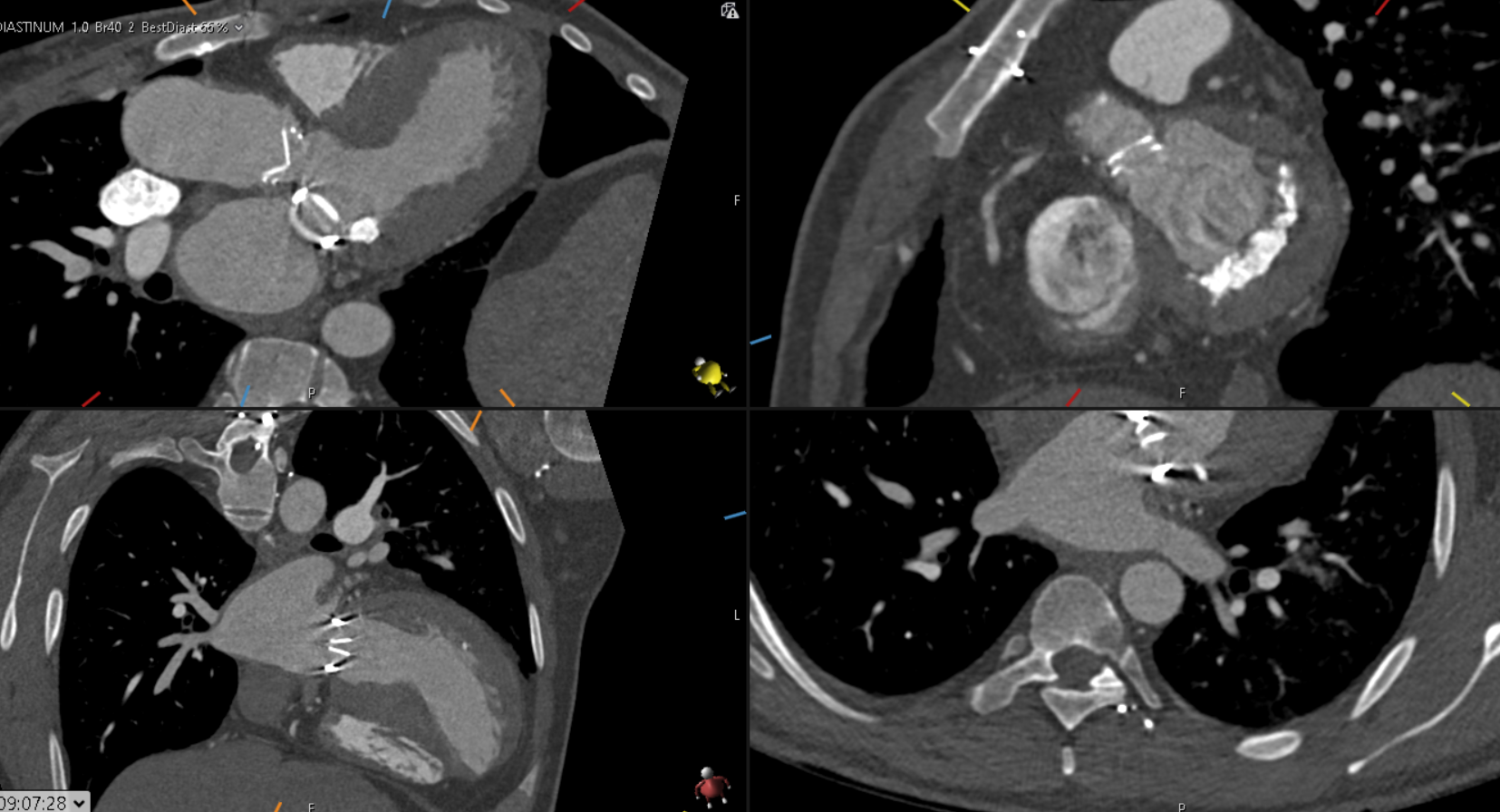
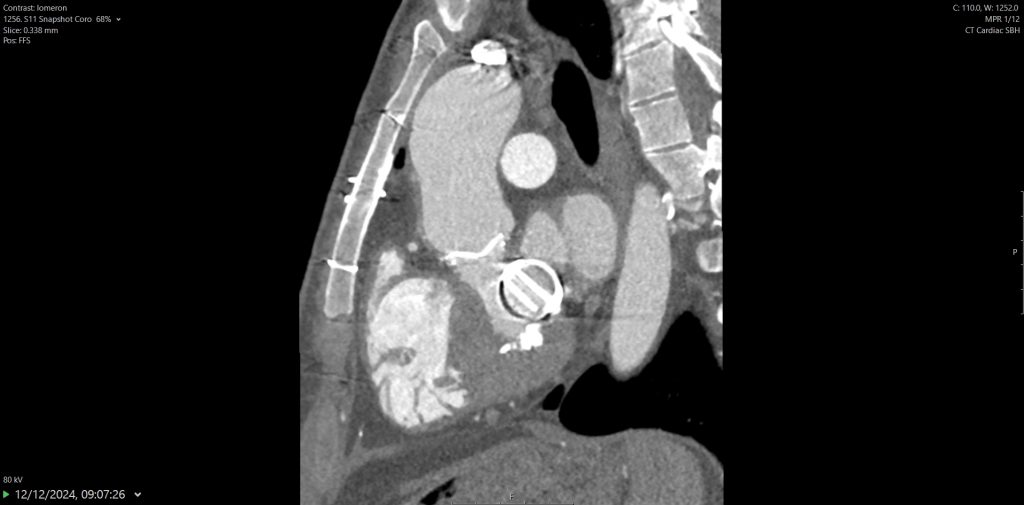
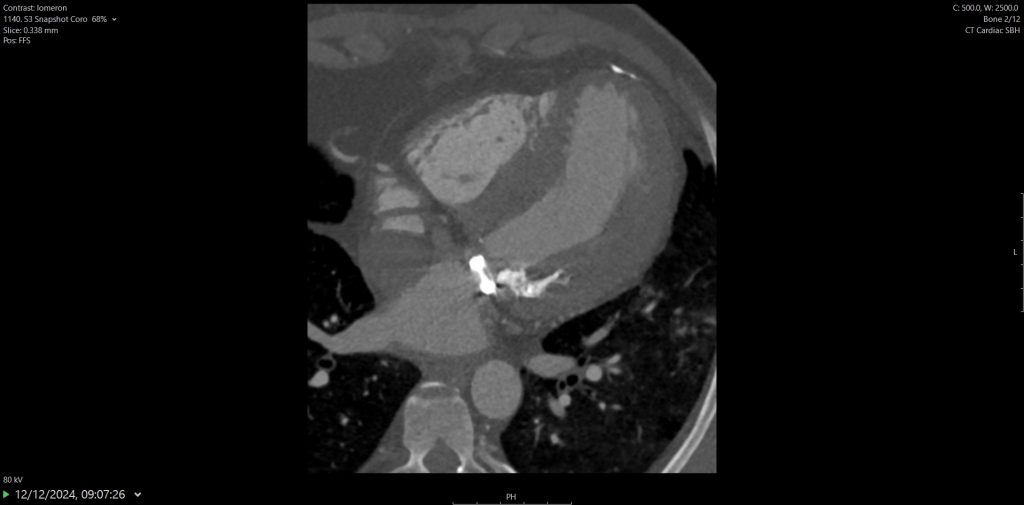
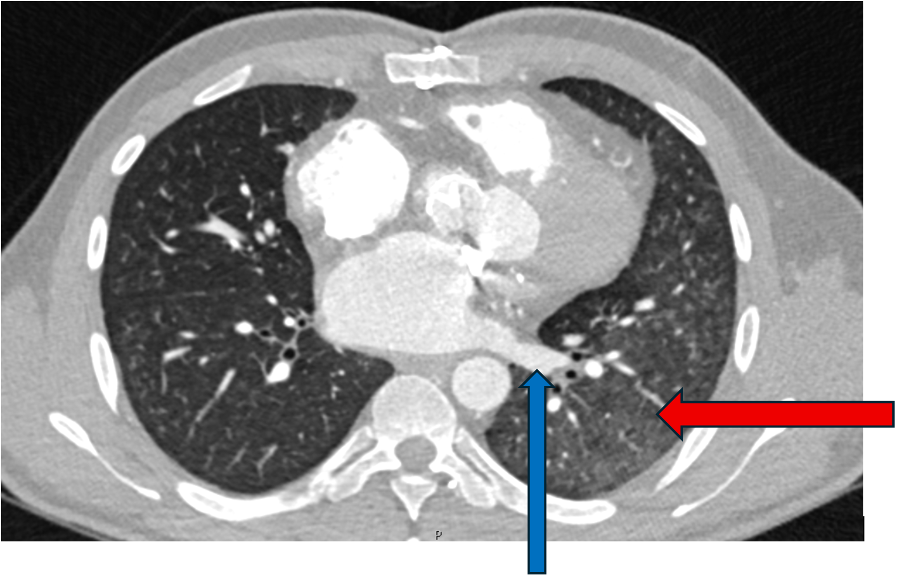
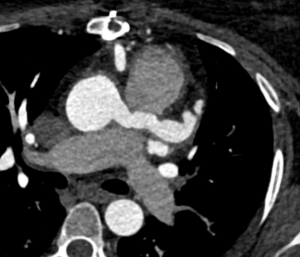
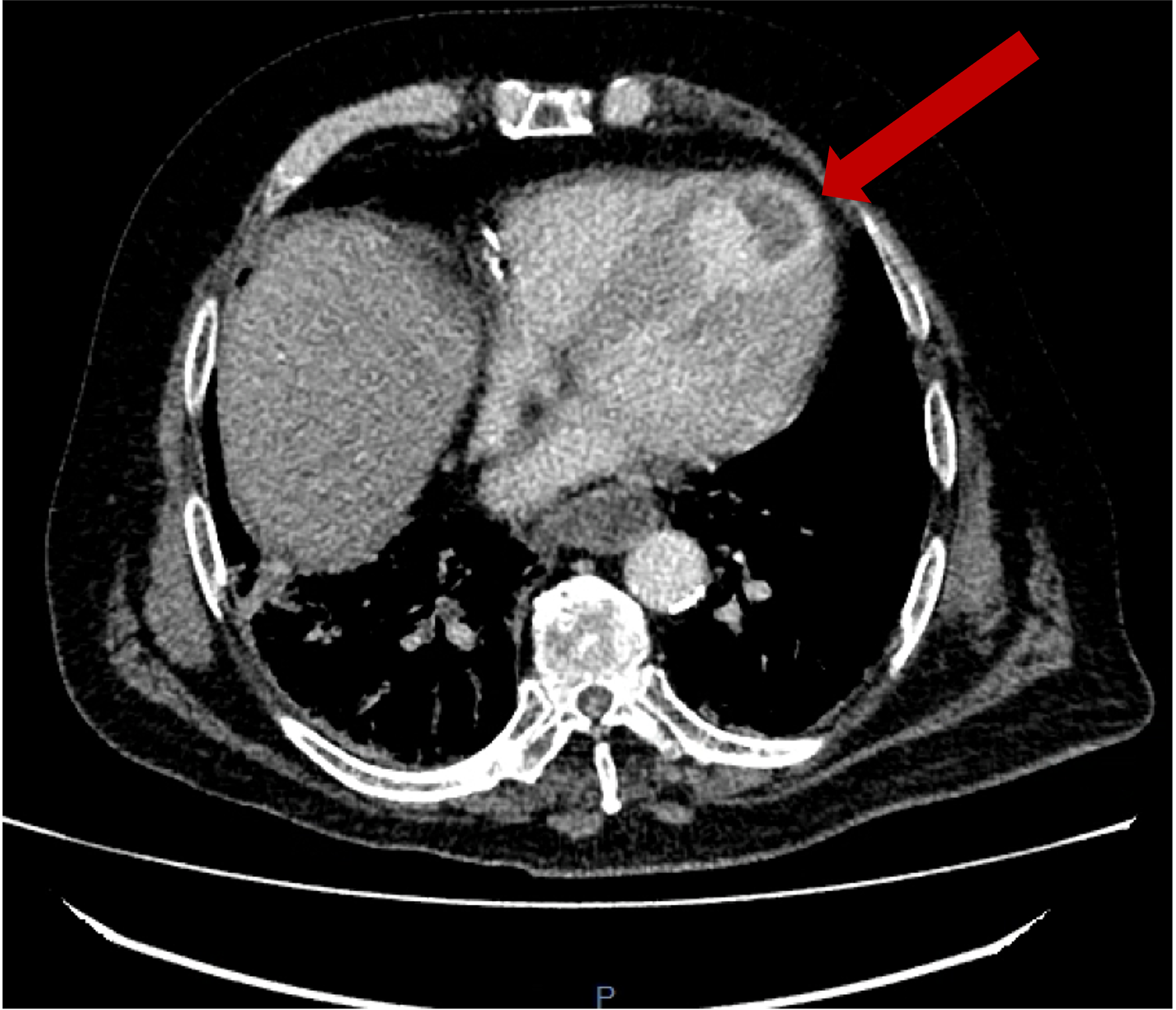
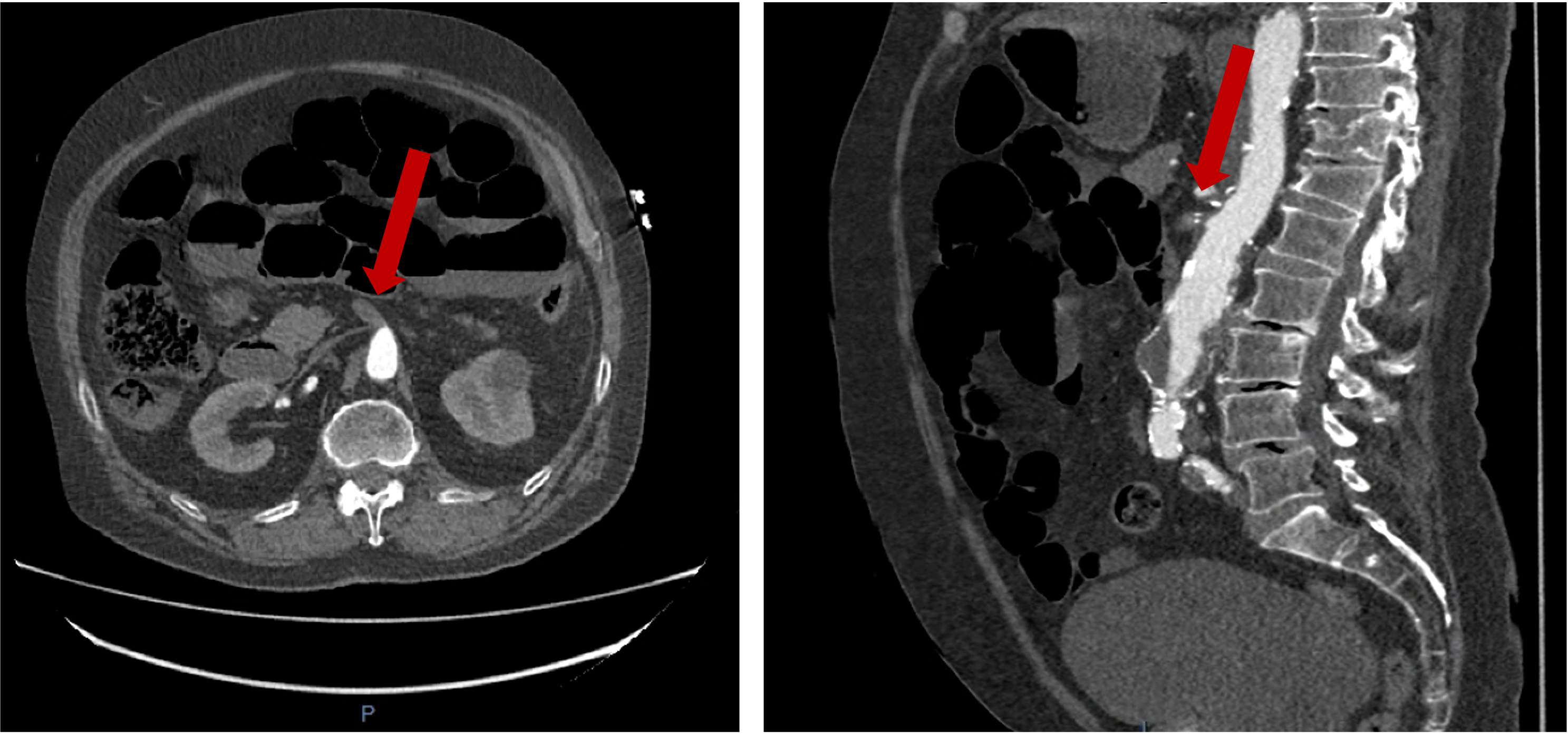
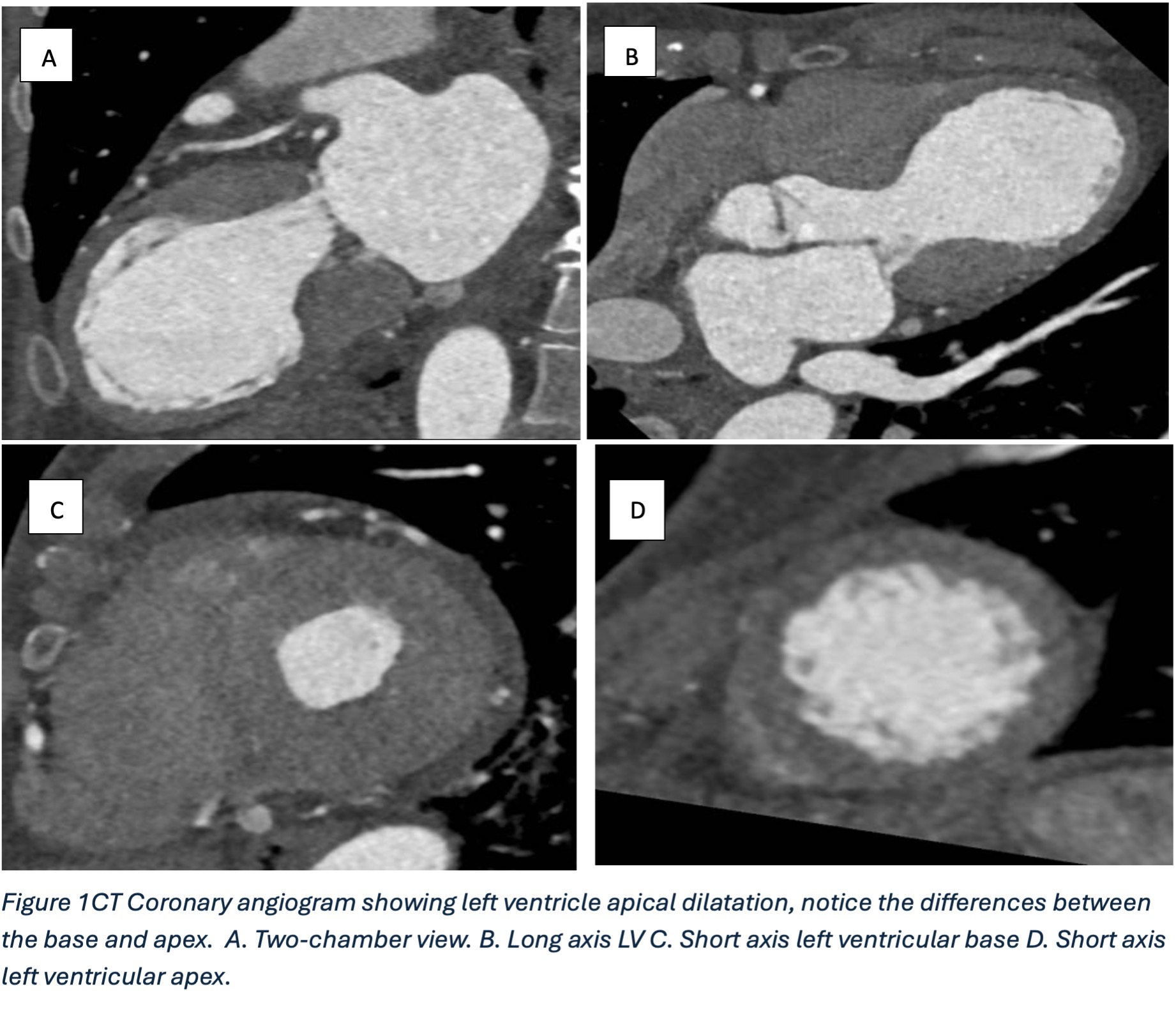
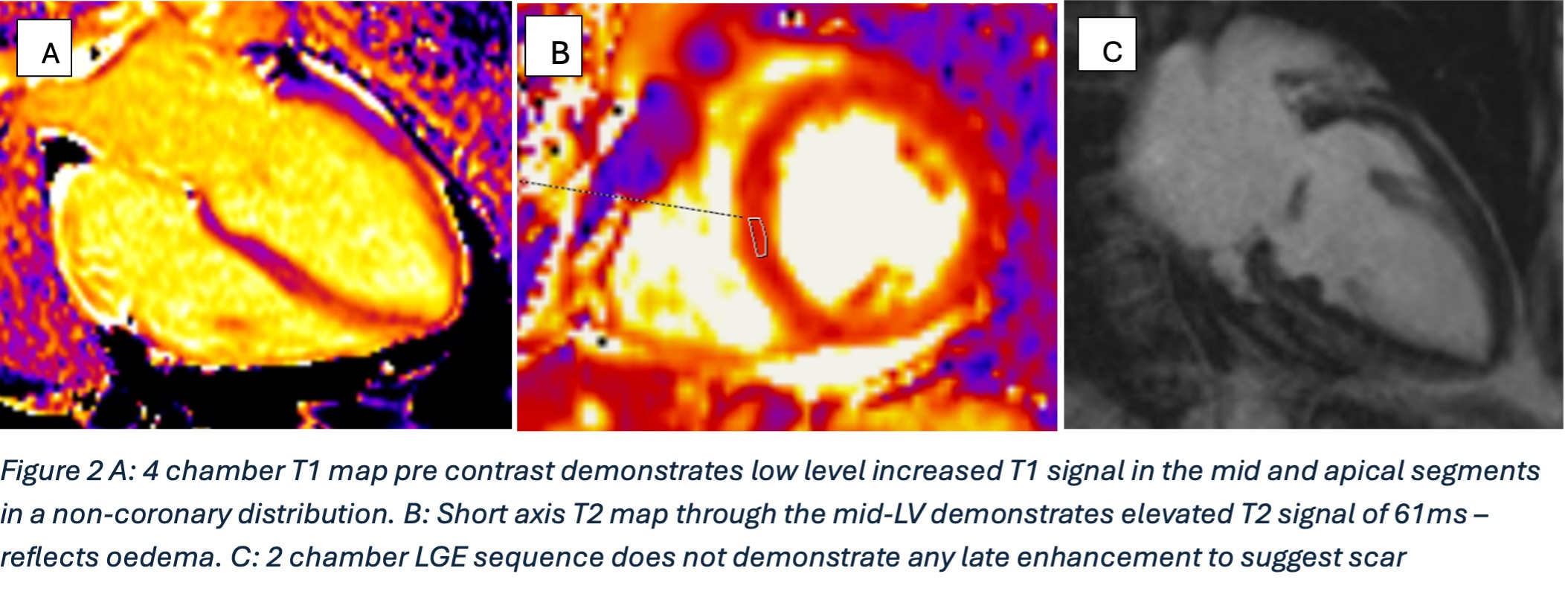
Surveillance CT demonstrated a filling defect at the left ventricular (LV) apex, consistent with thrombus, Figure 1. Coronal CT images additionally showed upper to mid zone ground-glass opacities and consolidation in the lungs bilaterally, a pattern that can be associated with eosinophilic pneumonia, Figure 2.
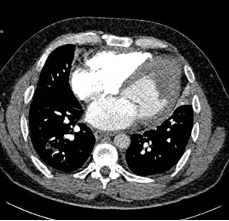
Figure 1
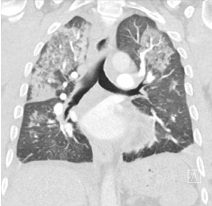
Figure 2
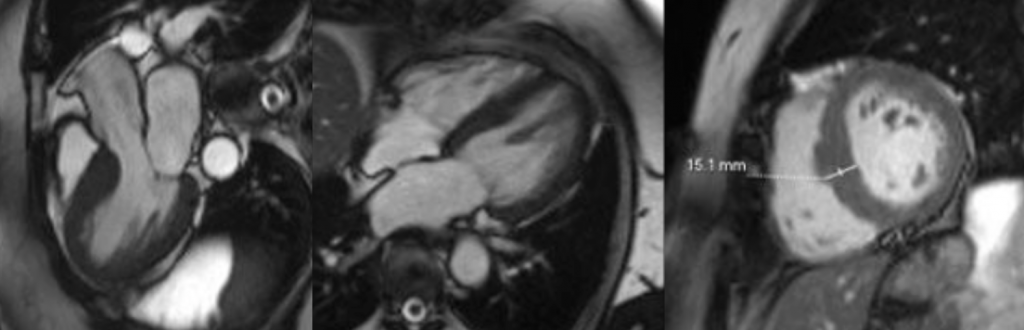
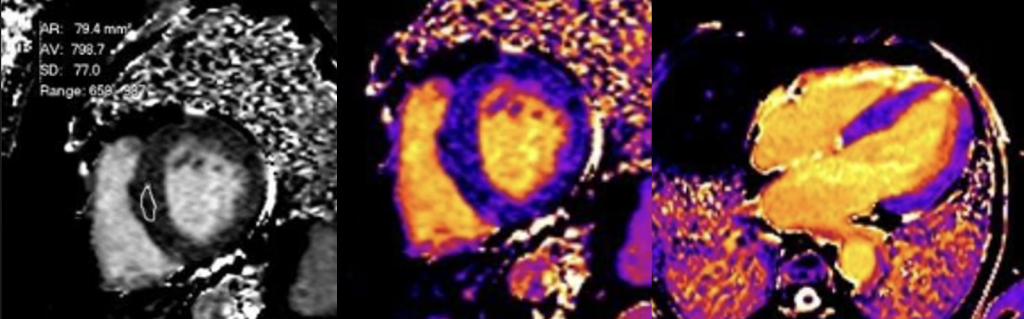
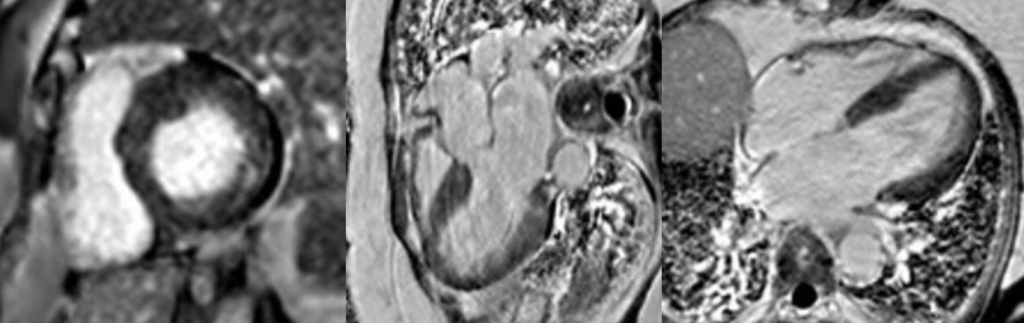
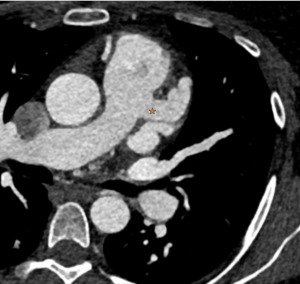
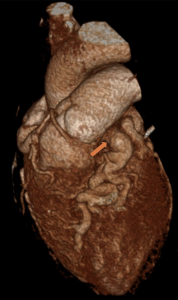
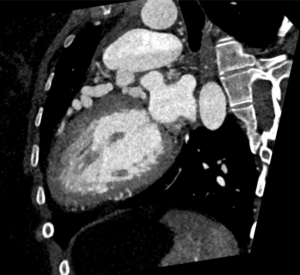
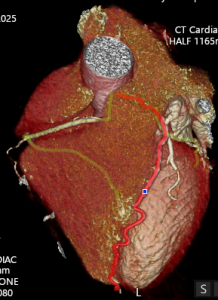
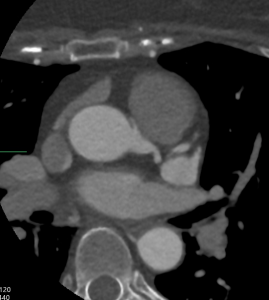
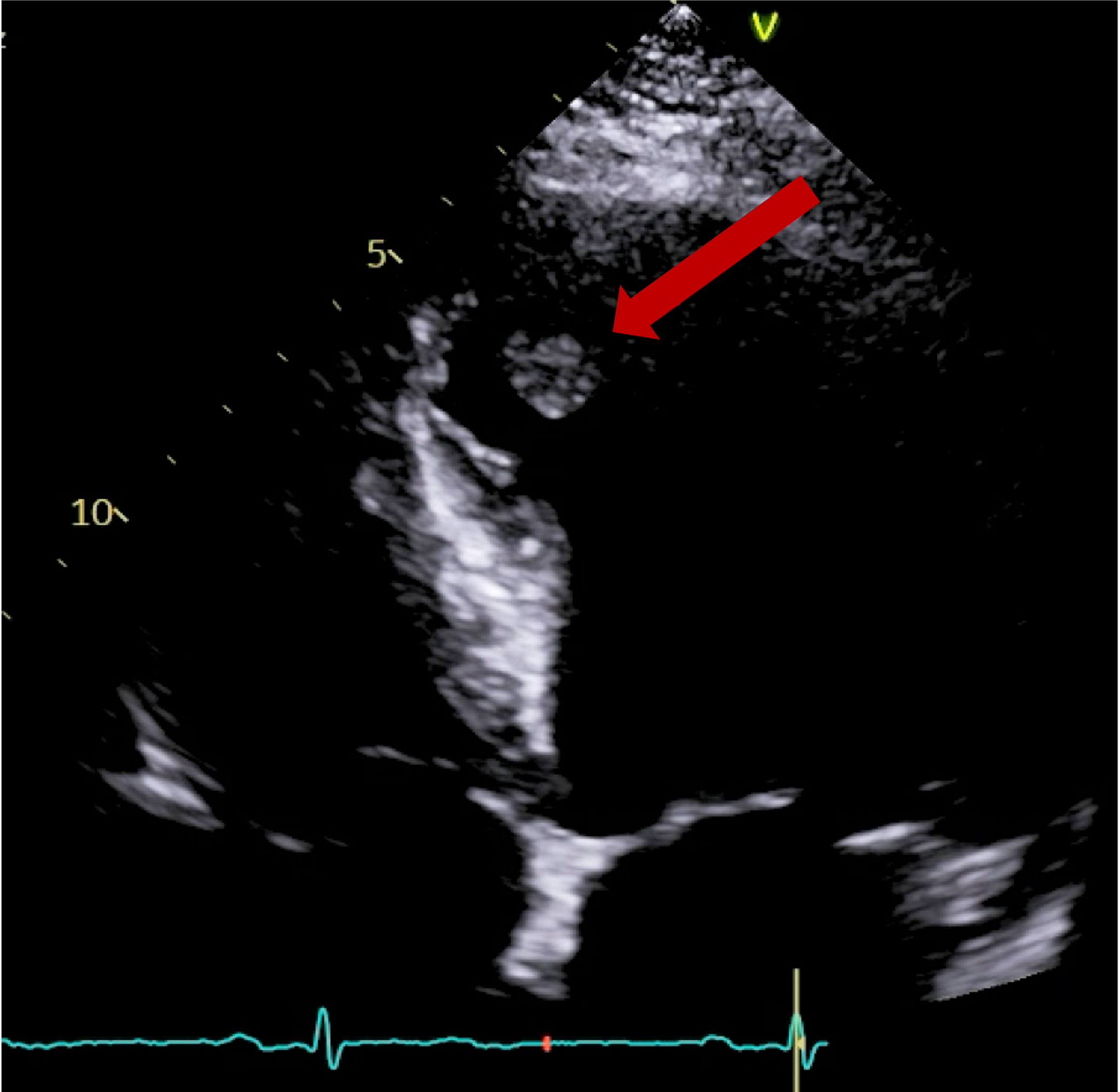
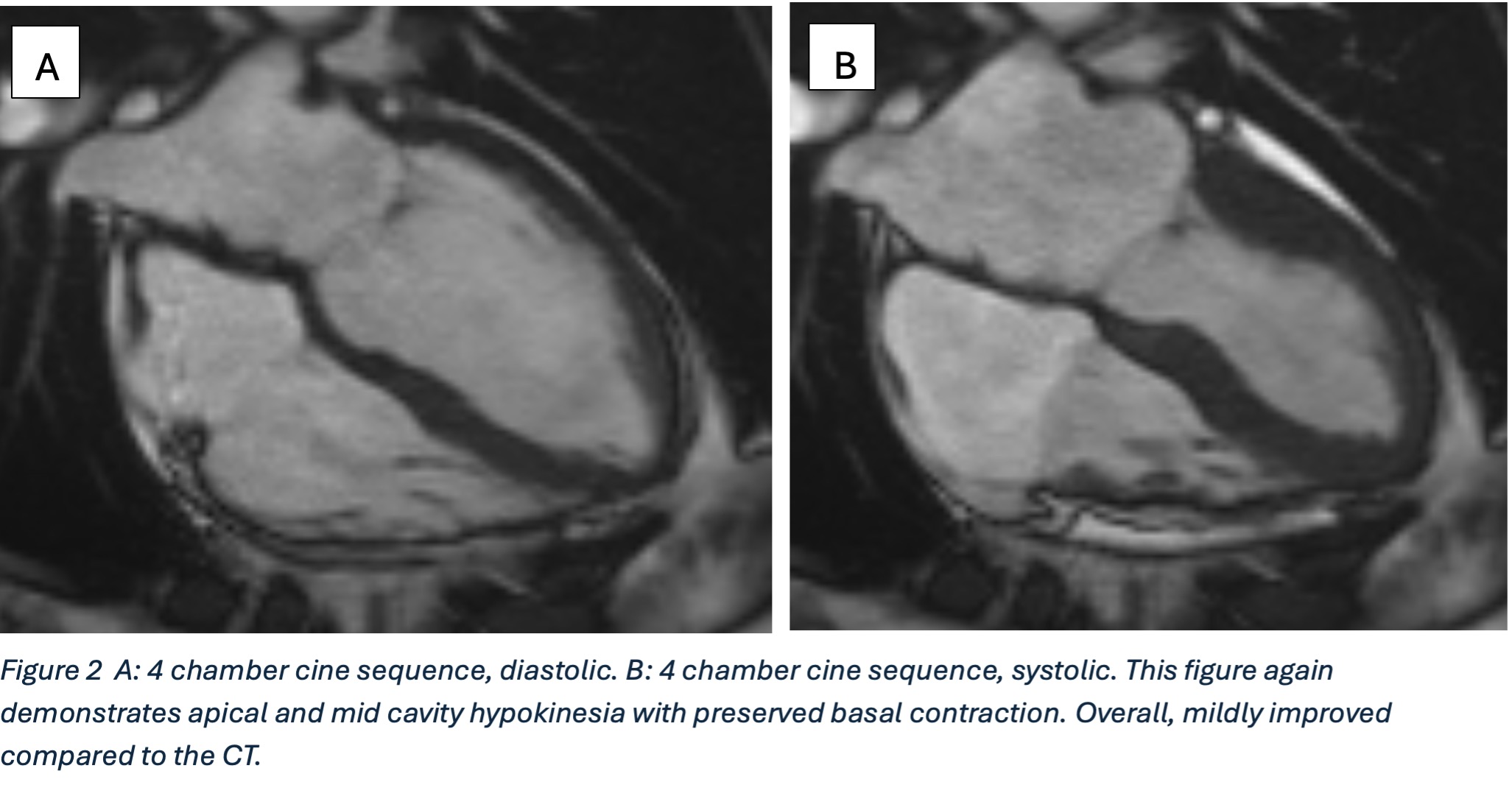
Cardiac MRI (CMR) revealed circumferential subendocardial late gadolinium enhancement (LGE) with an LV apical thrombus on delayed phase imaging, Figure 3. Four-chamber cine sequences showed relative hypokinesis involving the distal mid-to-apical segments, bi-atrial dilatation, and a right pleural effusion, Figure 4. These combined findings raised suspicion for restrictive cardiomyopathy, most consistent with eosinophilic myocarditis (EM).
Figure 3
Figure 4
Discussion
EM is a rare, heterogeneous inflammatory cardiomyopathy characterized by eosinophil-mediated myocardial injury that may progress through necrosis, thrombosis, and fibrosis, hence carrying a significant risk of morbidity and mortality.(1)
Idiopathic etiology is found to be the most common (28.8%), followed by eosinophilic granulomatosis with polyangiitis (19.3%), drug-induced (13.1%), and hypereosinophilic syndrome (12.8%), with the remaining cases linked to parasites, malignancy, or vaccines.(2)
While the classical “gold standard” for diagnosis is endomyocardial biopsy (EMB), noninvasive imaging, particularly CMR, has become central in raising suspicion for EM and guiding further evaluation.(3)
The combination of CT and MRI imaging findings in our patient — left ventricular (LV) apical thrombus, upper to mid zone ground-glass opacities and consolidation, circumferential subendocardial late gadolinium enhancement (LGE), regional wall motion abnormality (hypokinesis of mid-to-apical segments), and bi-atrial dilatation — aligns well with features described in histologically proven EM.
In a cohort of 15 biopsy-confirmed EM patients who underwent CMR, all showed myocardial edema. Most (93%) had multifocal subendocardial LGE, sometimes extending beyond the subendocardium. LV systolic dysfunction was common, even in non-dilated ventricles, and over half had pericardial or pleural effusions. Notably, two patients also had LV thrombi, similar to our case.(3)
EM can occur without peripheral eosinophilia, making diagnosis challenging.(4) Our patient was asymptomatic and undergoing routine cancer surveillance, so the diagnosis would likely have been missed without the incidental CT findings prompting further cardiac imaging. This case emphasises that recognising imaging features and maintaining a high index of suspicion are key to timely diagnosis. However, EMB remains the gold standard for diagnosis.
Management:
EM is rare, and no large-scale studies define standard management. First-line therapy is high-dose corticosteroids to suppress myocardial injury and prevent fibrosis.(5) Anticoagulation is indicated for LV thrombus to reduce embolic risk.(6) Identifying underlying causes—such as hypersensitivity, HES, or parasitic infection—is important to guide targeted therapy.(7) In steroid-refractory or relapsing cases, IL-5–targeted biologics (e.g., mepolizumab, benralizumab) may be beneficial.(5) Long-term CMR follow-up helps monitor inflammation, resolution, and functional recovery.(3)
Multiple-Choice Questions
- Which CMR feature in this case is most characteristic of eosinophilic myocarditis?
A. Transmural LGE in a coronary artery distribution
B. Circumferential subendocardial LGE
C. Mid-wall septal enhancement only
D. No enhancement with preserved T2 signal
E. Patchy epicardial enhancement - The presence of an apical LV thrombus in EM is most likely due to:
A. Primary hypercoagulability
B. Coronary artery embolism
C. Endocardial damage in the necrotic stage
D. Systemic hypotension
E. Pericarditis - Which finding suggests evolving restrictive physiology?
A. Hyperdynamic LV function
B. Apical ballooning
C. Bi-atrial dilatation with apical hypokinesis
D. Global RV dilation
E. Pericardial calcification - First-line therapy for acute EM is:
A. Beta-blockers
B. High-dose corticosteroids
C. IL-5 inhibitors
D. Diuretics only
E. Surgical resection
Answers: B, C, C, B
References
- Zaid Ammouri, Sami Belkouchia, Ibtissam Rezzouk, Salma Moussaoui, & Habbal, R. (2024). Eosinophilic Myocarditis: A Diagnostic Challenge and Treatment Dilemma – A Case Report. European Heart Journal – Case Reports, 8(10). https://doi.org/10.1093/ehjcr/ytae418
- Techasatian, W., Gozun, M., Vo, K., Yokoyama, J., Nagamine, T., Shah, P., Vu, K., Zhang, J., & Nishimura, Y. (2023). Eosinophilic myocarditis: systematic review. Heart. https://doi.org/10.1136/heartjnl-2023-323225
- Pauli Pöyhönen, Rågback, J., Mäyränpää, M. I., Hanna-Kaisa Nordenswan, Lehtonen, J., Shenoy, C., & Markku Kupari. (2023). Cardiac magnetic resonance in histologically proven eosinophilic myocarditis. Journal of Cardiovascular Magnetic Resonance, 25(1), 79–79. https://doi.org/10.1186/s12968-023-00979-0
- Lee, J.-Y., Sun Hwa Lee, & Won Ho Kim. (2023). Fulminant Eosinophilic Myocarditis Without Peripheral Eosinophilia. Texas Heart Institute Journal, 50(2). https://doi.org/10.14503/thij-21-7818
- Asada, A. M., Kahwash, R., & Trovato, V. (2025). Eosinophilic Myocarditis: A Concise Review. Current Cardiology Reports, 27(1). https://doi.org/10.1007/s11886-024-02184-6
- Brambatti, M., Matassini, M. V., Adler, E. D., Klingel, K., Camici, P. G., & Ammirati, E. (2017). Eosinophilic Myocarditis: Characteristics, Treatment, and Outcomes. Journal of the American College of Cardiology, 70(19), 2363–2375. https://doi.org/10.1016/j.jacc.2017.09.023 org/10.1016/j.jacc.2017.09.023
- Schulz-Menger, J., Collini, V., Gröschel, J., Adler, Y., Brucato, A., Christian, V., Ferreira, V. M., Gandjbakhch, E., Heidecker, B., Kerneis, M., Klein, A. L., Klingel, K., Lazaros, G., Lorusso, R., Nesukay, E. G., Rahimi, K., Ristić, A. D., Rucinski, M., Sade, L. E., & Schaubroeck, H. (2025). 2025 ESC Guidelines for the management of myocarditis and pericarditis. European Heart Journal. https://doi.org/10.1093/eurheartj/ehaf192