Authors:
Dr Simon Woodbridge1, Dr Giulia Benedetti1
Affiliations:
1Guy’s & St Thomas’ NHS Foundation Trust
Case Presentation:
A 65 year old male presented following a 6 month history of progressively worsening exertional angina, on a background of unremarkable preliminary cardiac investigations, including normal electrocardiography. There was no history of exertional breathlessness or syncope. Other symptoms included occasional dizziness unrelated to posture or position, spontaneous shoulder cramping/stiffness, and occasional unilateral right sided headaches which would subside after approximately 1 hour.
His background medical history included previous tonsillar and prostate cancer, both in remission, and aside from being an ex-smoker, he otherwise demonstrated no risk factors for cardiovascular disease.
Imaging Findings:
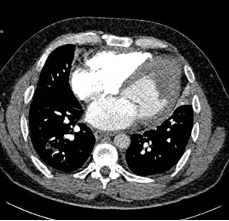
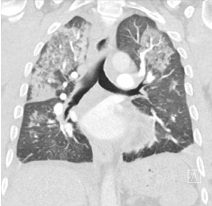
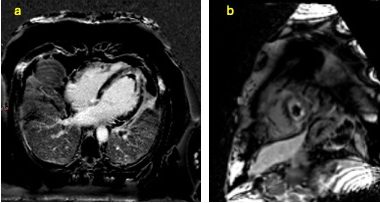
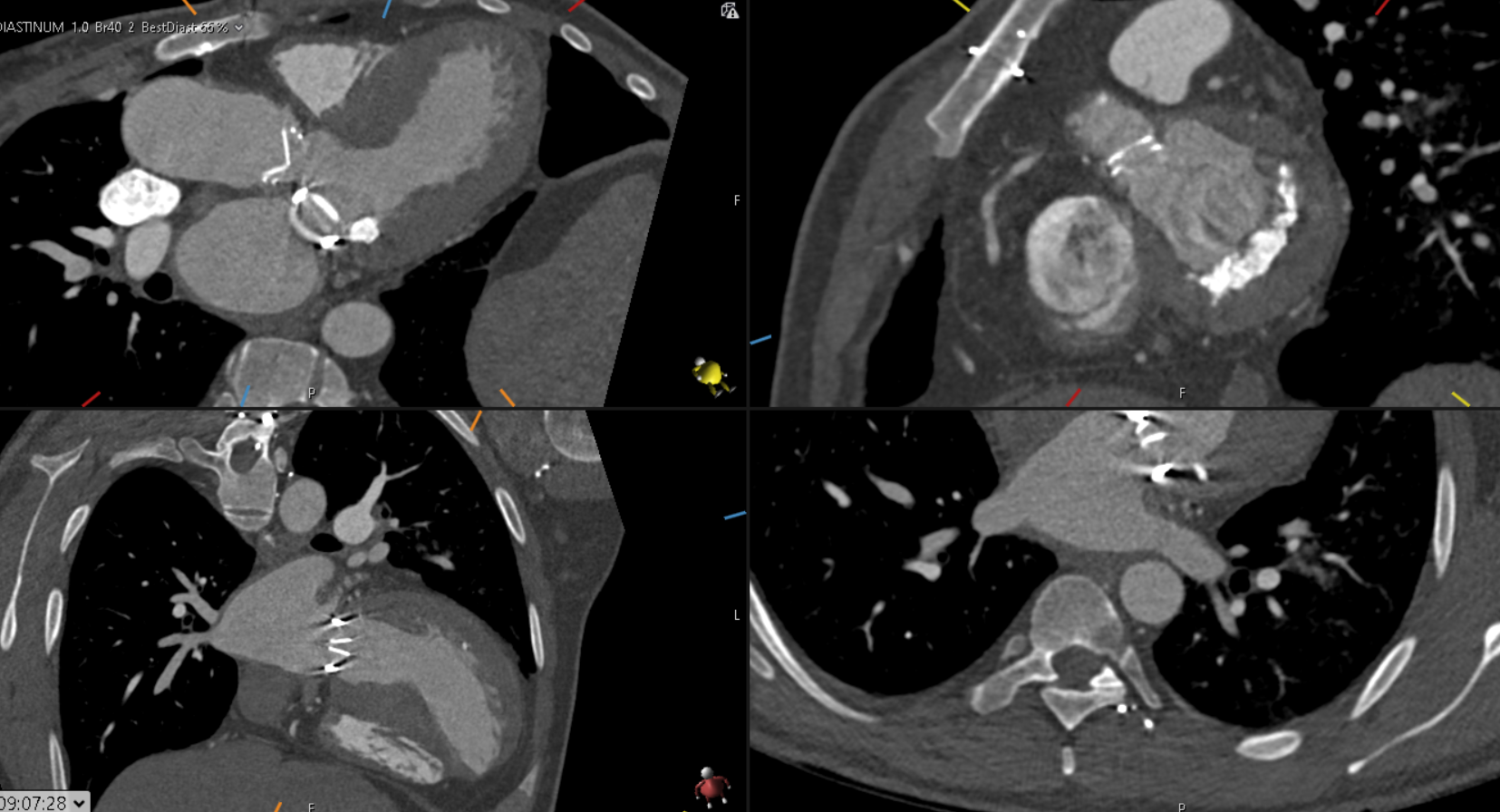
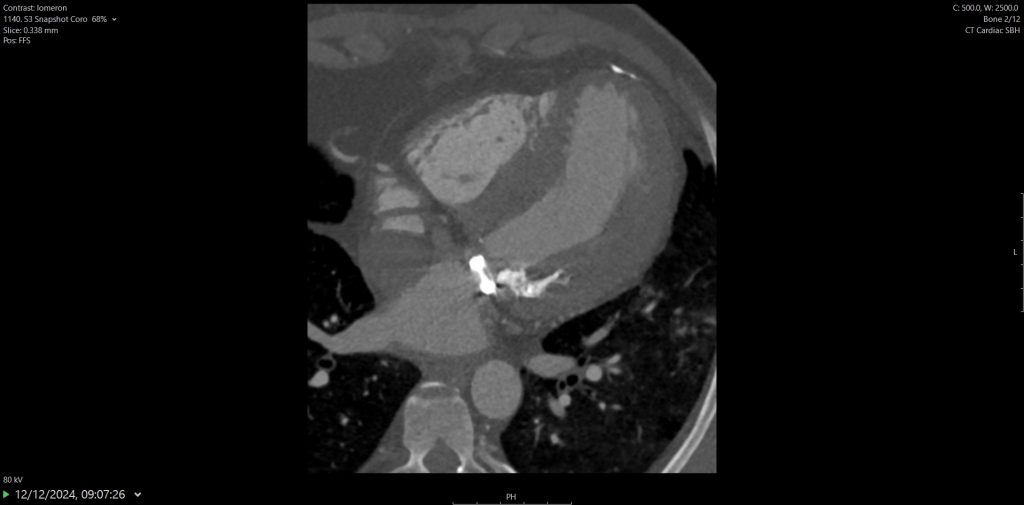
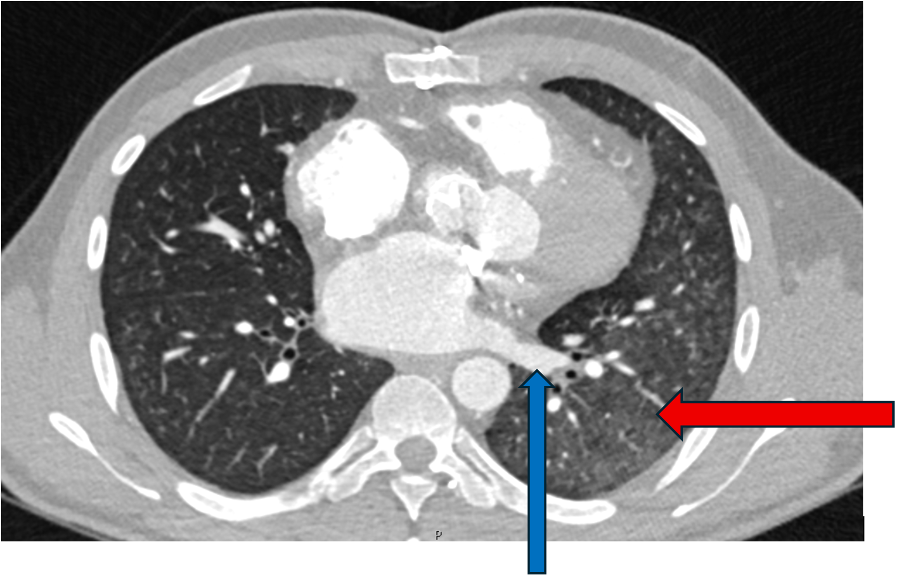
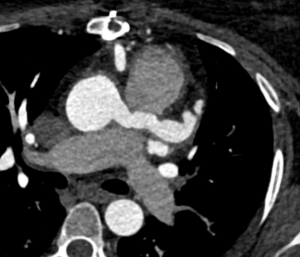
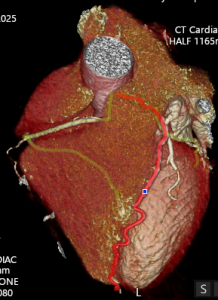
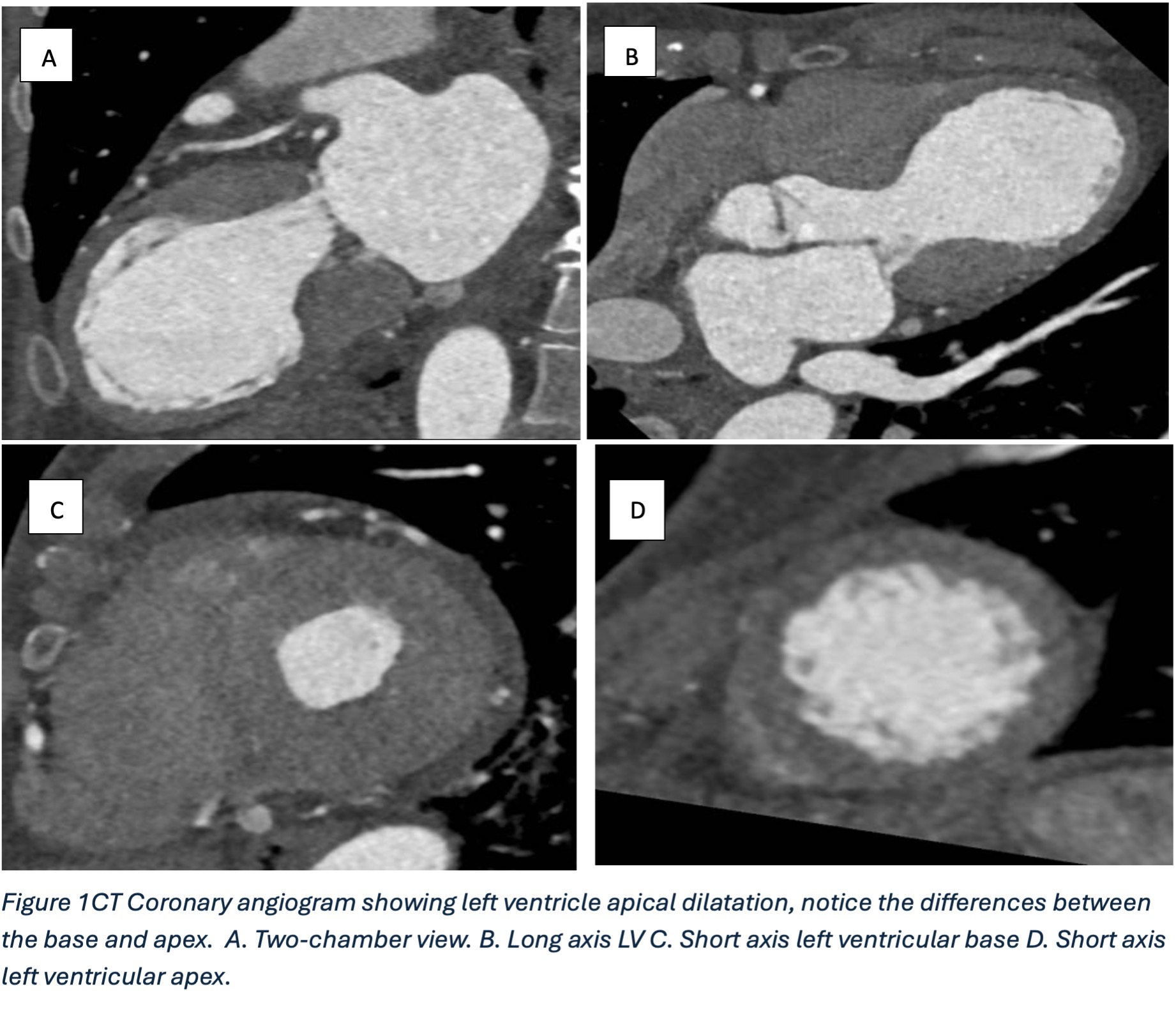
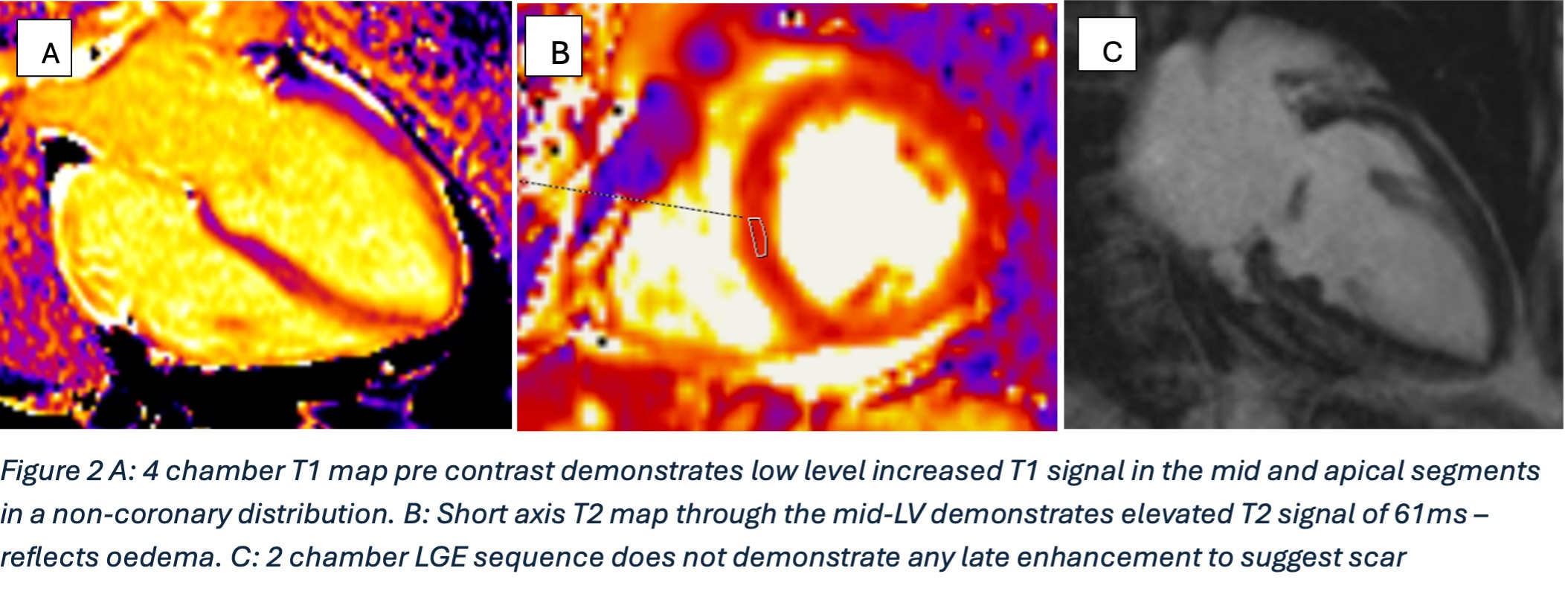
He was referred for CT coronary angiography to further investigate (Figure 1). This study revealed largely concentric perivascular soft tissue thickening affecting the left main stem, proximal left anterior descending (LAD) and left circumflex arteries, and proximal right coronary artery. At the affected areas, there was overall moderate (50-69%) stenosis. There was minimal calcific plaque within the proximal RCA.
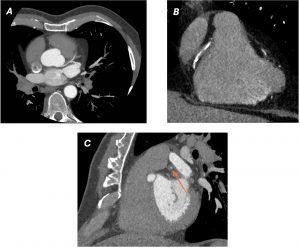
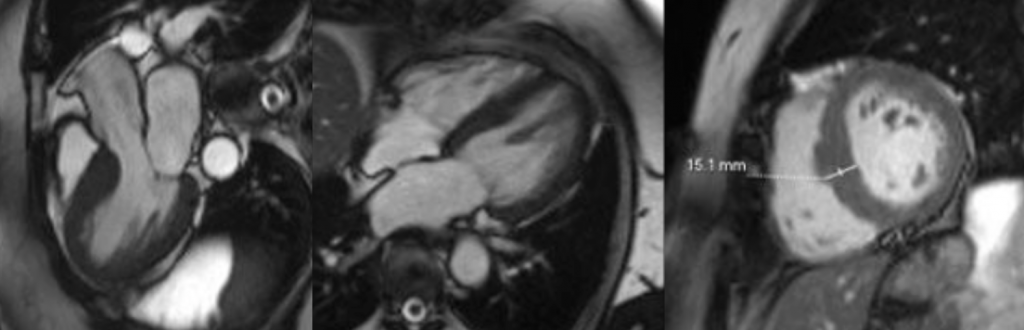
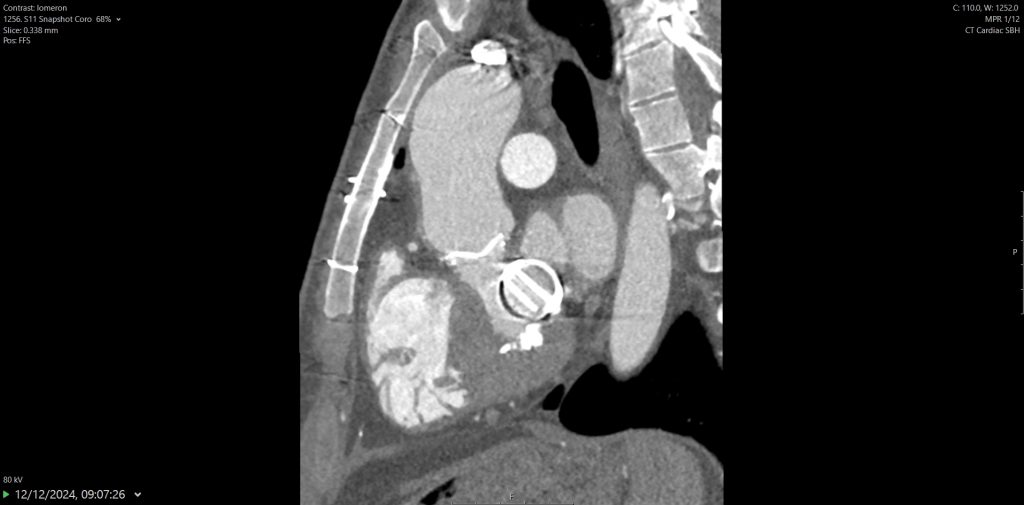
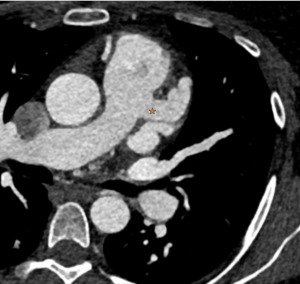
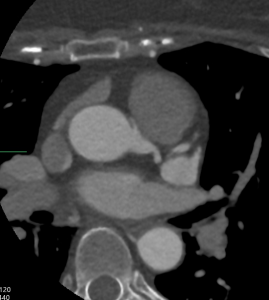
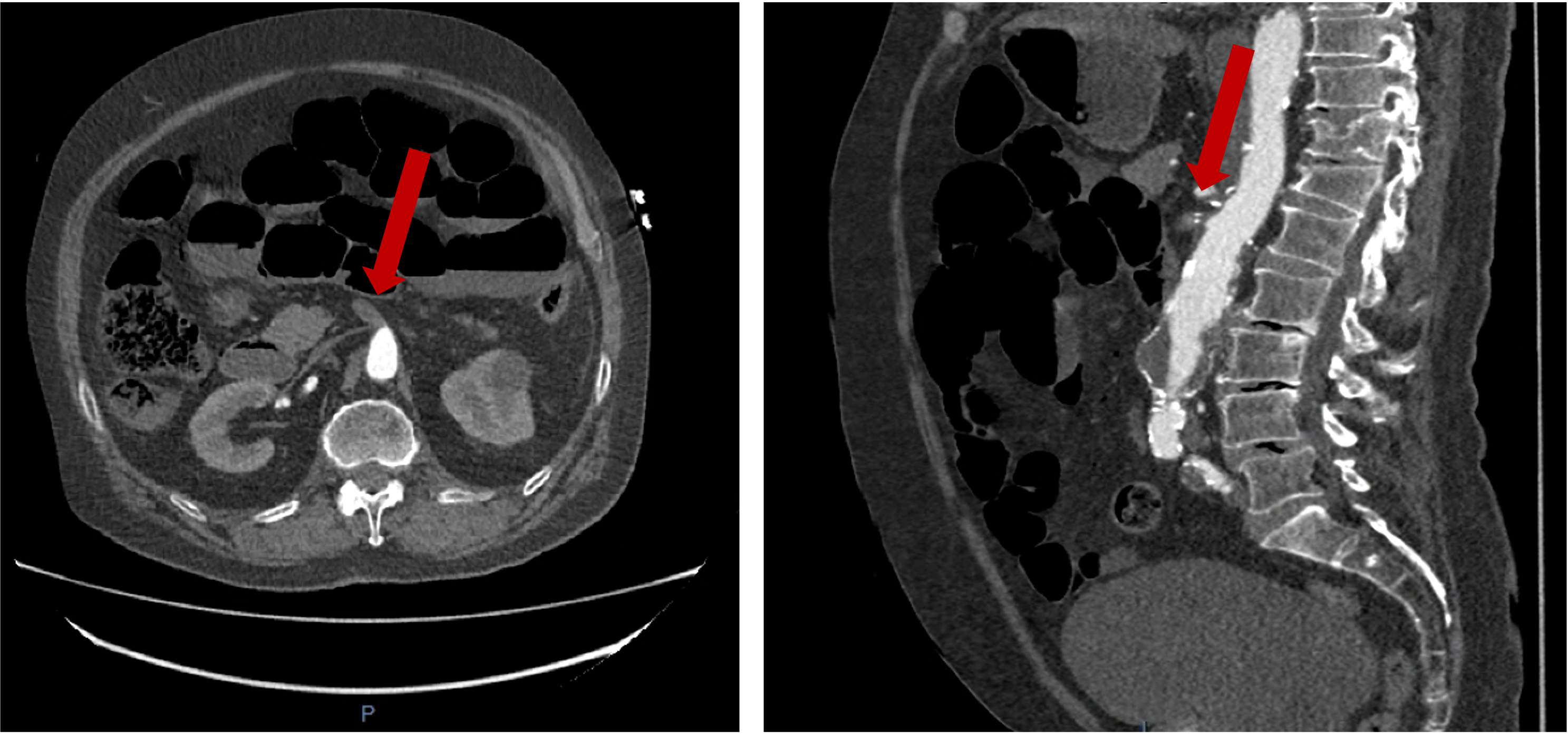
Due to the perivascular morphology of soft tissue thickening, rather than intraluminal plaque causing stenosis, in addition to the distribution of the abnormality predominantly within the proximal coronary vasculature, the possibility of vasculitis was raised, and further work-up was performed with a CT aortic angiogram (Figure 2). This study revealed circumferential aortic soft tissue thickening within the aortic arch, abdominal aorta, and proximal common iliac arteries. The remainder of the major aortic branch arteries, including the renal arteries, were spared. Suspicion of a vasculitic process was therefore increased.
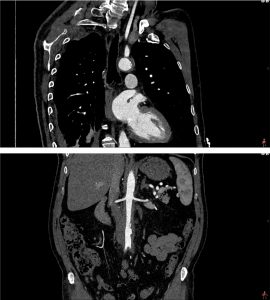
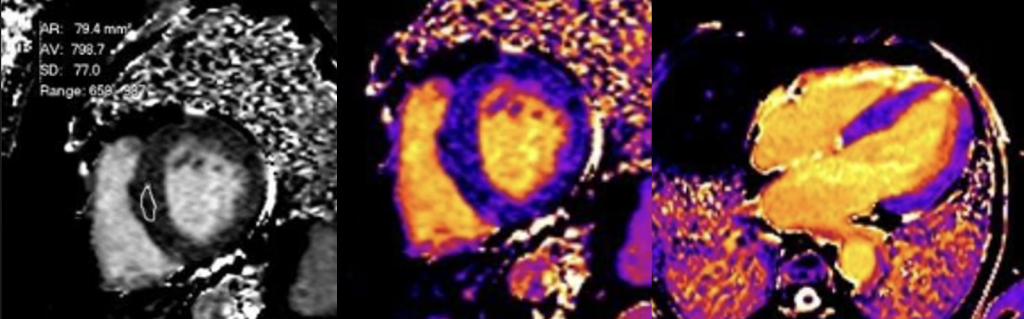
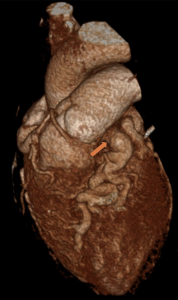
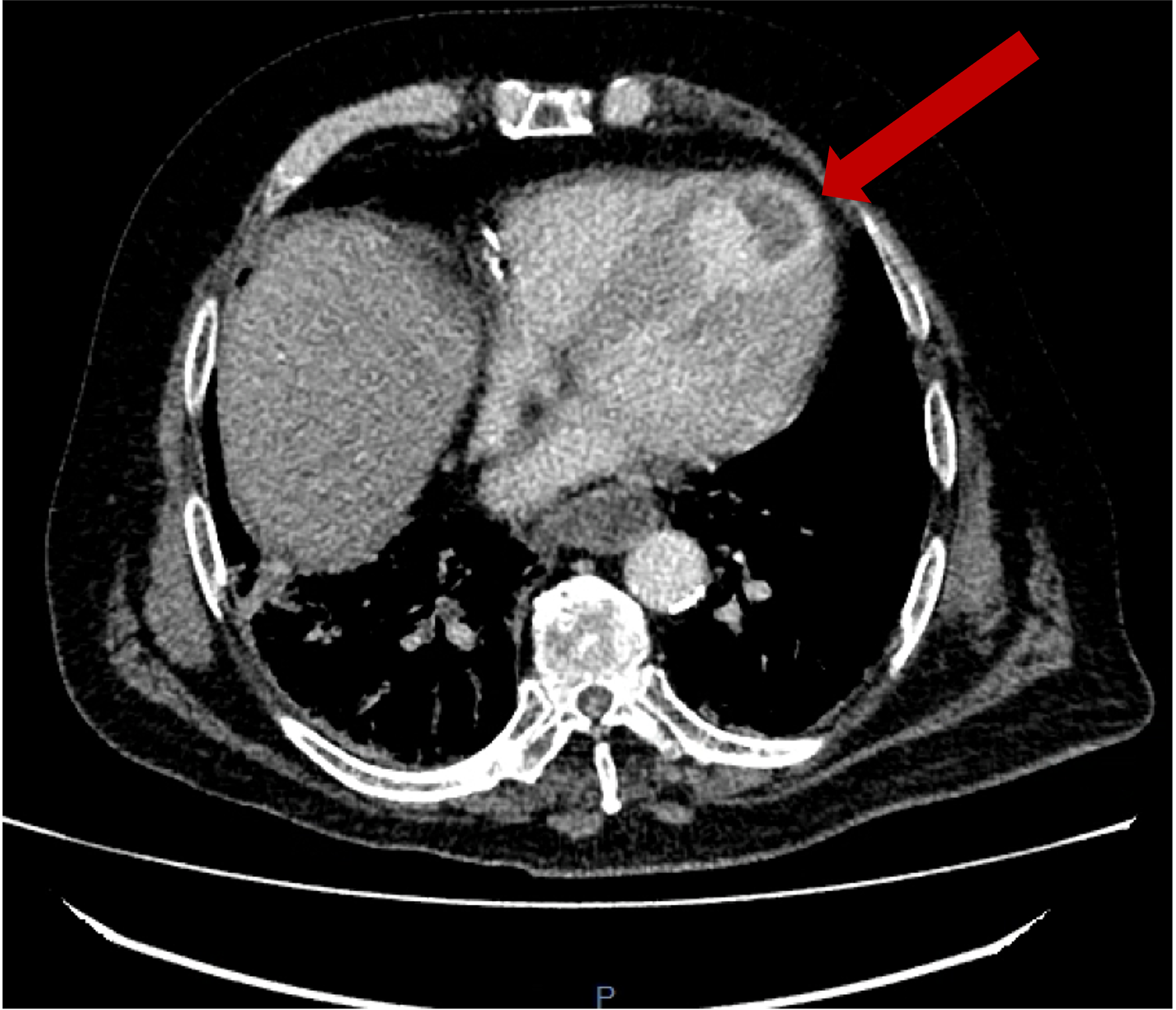
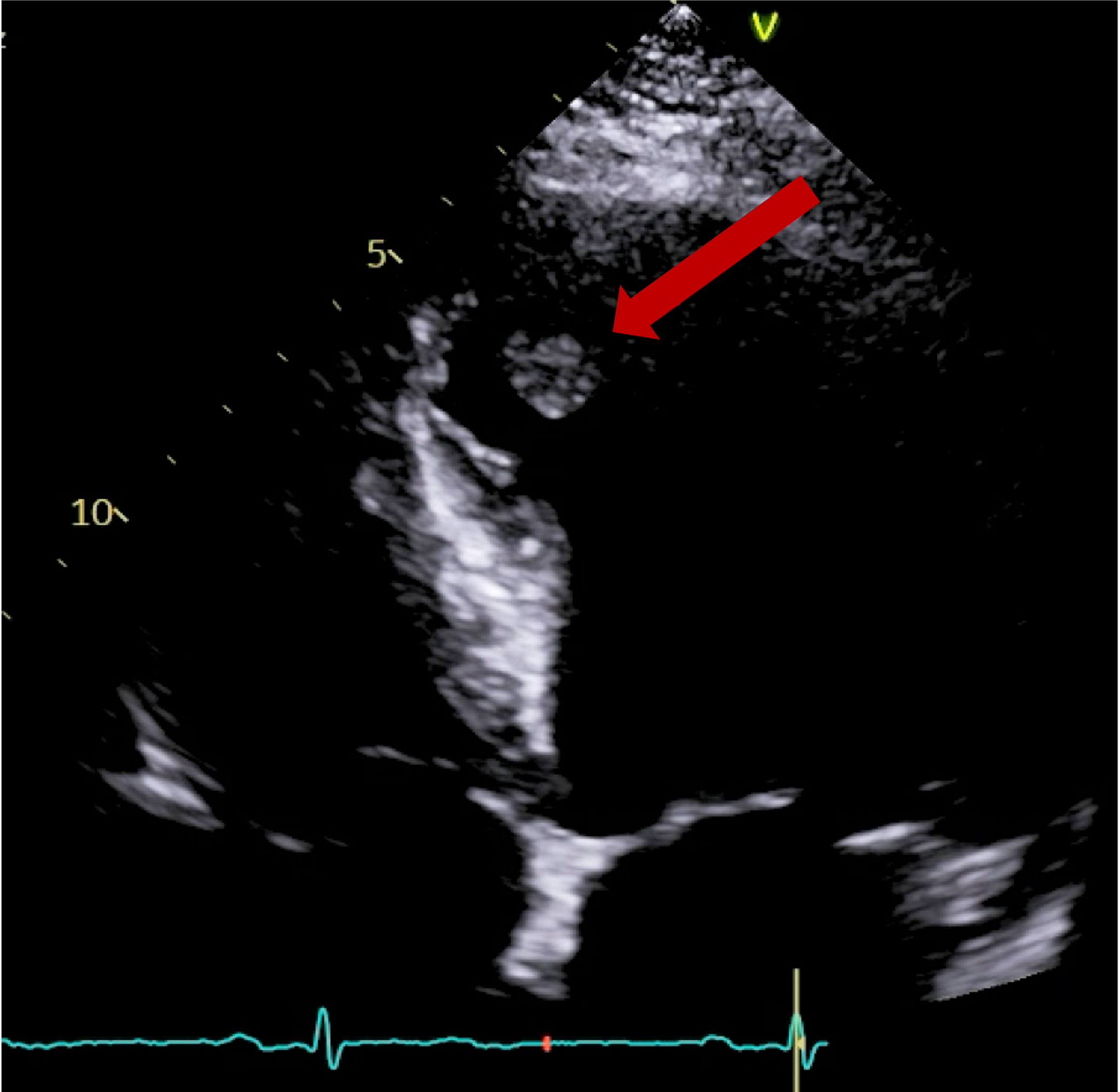
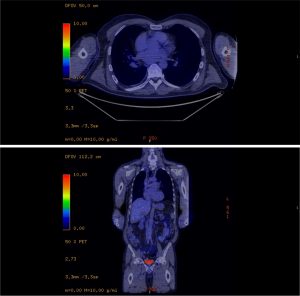
A subsequent FDG PET/CT study was performed (Figure 3). The PET/CT study demonstrated avid tracer uptake within the aorta and common iliac arteries at the sites of soft tissue thickening demonstrated on the prior CT aortic angiogram. In addition, a linear region of uptake correlating with the peri-coronary soft tissue along the LMS/LAD was identified, allowing for slight anatomical misalignment artefact and myocardial glucose utilisation. The distribution of inflammation, primarily centred around the coronary arteries, aorta and common iliac arteries, was radiologically deemed most in keeping with an IgG4 related vasculitis.
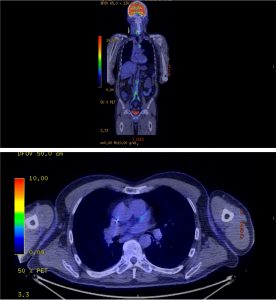
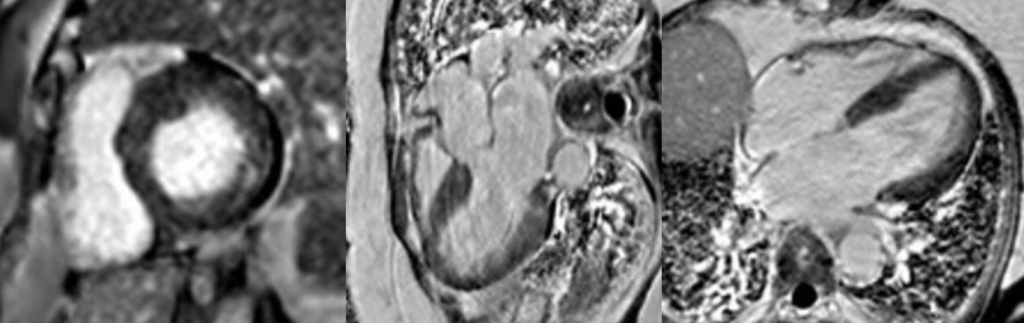
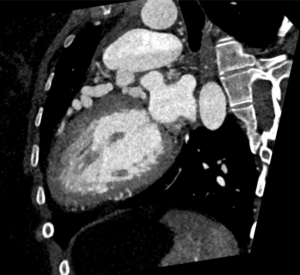
The patient was treated as a large vessel vasculitis, secondary to probable IgG4-related disease (serum IgG4 levels, reportedly not demonstrating high specificity in this condition, were normal) and commenced on high dose corticosteroid treatment. A repeat FDG PET/CT study was performed 1 month following treatment (Figure 4), which demonstrated a dramatic improvement, with minimal periaortic tracer uptake in the previously affected areas, in addition to complete cessation of uptake within the coronary arteries.
Discussion:
IgG4 related disease (IgG4-RD) is a rare autoimmune systemic disease which is characterised by fibroinflammatory infiltration of organs by plasma cells expressing IgG4 antibodies. Elevated serum IgG levels are seen in 70-80% of patients, hence, normal levels do not exclude the disease.
There is a broad disease spectrum encompassing nearly all body systems and cardiovascular involvement can include aortitis, peri-aortitis, arteritis and coronary arteritis within its phenotype. In addition to coronary arterial and aortic inflammation, involvement of the common and external iliac arteries is the next most common affected vasculature, which was also exhibited in this patient.
CTCA, CTA and PET/CT imaging are key investigations which aid the diagnosis of IgG4 related cardiovascular disease. In CTA an additional delayed phase can assist in differentiating inflammatory perivascular thickening from mural thrombus formation if there is uncertainty in interpretation, showing possible contrast uptake within the soft tissue. In addition to diagnosis, PET/CT may also be a primary contributor in monitoring disease activity, as it is possible (as with this patient) that biochemical markers of inflammation do not correlate with disease extent.
Corticosteroids are the primary treatment of IgG4-RD, supplemented with methotrexate or mycophenolate mofetil/azathioprine as a second line option if there is limited response. A third line treatment option is B-cell depletion with Rituximab. This patient demonstrated a significant radiological and clinical response to corticosteroid treatment.
References:
Sudheer, N., Oliver, T.I. (2019). IgG4 Related Disease (IgG4-RD). [online] Nih.gov. Available at: https://www.ncbi.nlm.nih.gov/books/NBK499825/
Vasculitis UK. (n.d.). Immunoglobulin G4 Disease (IgG4-RD). [online] Available at: https://www.vasculitis.org.uk/about-vasculitis/immunoglobulin-g4
Oyama-Manabe, N., Satoshi, Y., Manabe, O., Kato, F., Hiromi Kanno-Okada and Kudo, K. (2018). IgG4-related Cardiovascular Disease from the Aorta to the Coronary Arteries: Multidetector CT and PET/CT. Radiographics, 38(7), pp.1934–1948. doi:https://doi.org/10.1148/rg.2018180049